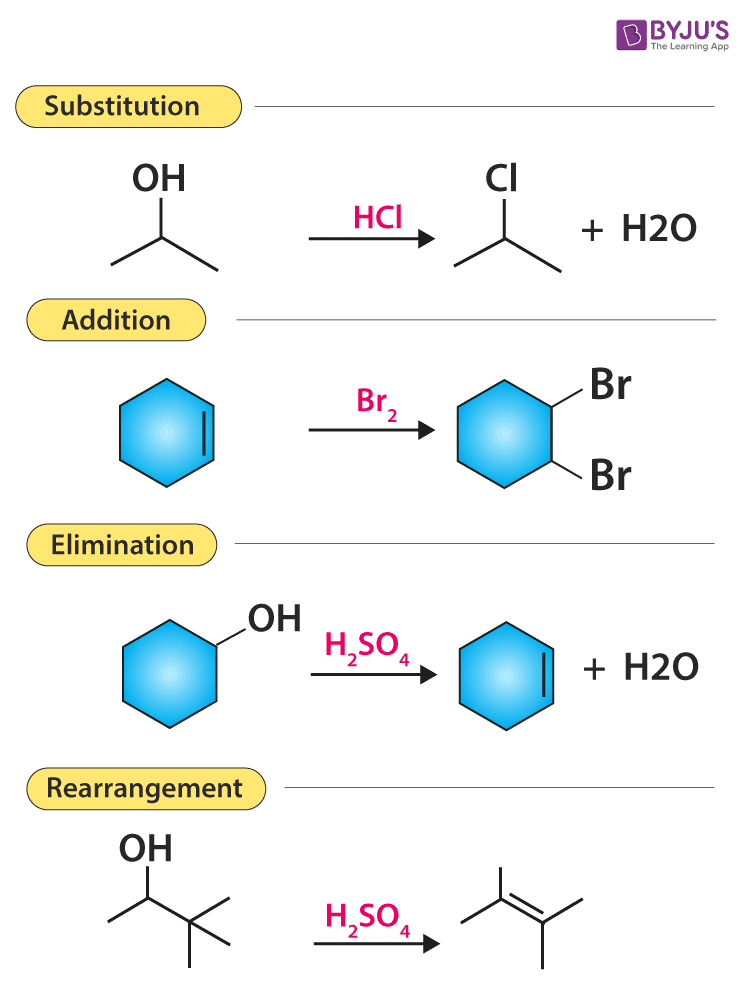
In this particular reaction 32.0 grams of methane burns completely to produce 66.0 grams of water. Let's do some examples of simple reactions.Ĭonsider the combustion of methane to form carbon dioxide and water. Percentage yield is calculated from the mass of reactants and the mass of products. Where atom economy proves useful, however, is in comparing different reaction pathways to a desired product.Ītom economy is the percentage of the total mass of reactants that successfully converted to the desired product. 
Atom economy does not take into account yield and does not allow for the fact that many industrial processes deliberately use excess of reactants to increase the yield of a required product. Percentage yield and atom economy can be calculated from the balanced chemical equation prior to any reaction taking place. Atom economy on the other hance, tells us how many atoms in the total number pf reactant atoms will be converted into our desired product. Percentage yield tells us the amount of the reactants that should react to form the product have successfully converted into products. In other words, wasteful a reaction is. It gives and indication of the amount of waste products produced during a reaction in forming a desired product. Percentage atom economy is really a measure of the sustainability of a chemical reaction. In other words, atom economy is a calculation which measures "how much of the reactant atoms actually form the final product", the higher the atom economy the lower the amount of waste product formed. The ideal atom economy, known as 100% atom economy, for a chemical reaction is taken as the process where all reactant atoms are found in the desired product. 
Atom economy is the second principle of green chemistry Chemistry-atom economy and percentage yield Atom economy and percentage yield.Ītom economy and percentage yield are indicators of how efficient a chemical reaction is.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
